Guidance for OJJDP Research Grantees
All OJJDP awards that include research activities and those made under funding opportunities funded with OJJDP research funds have additional research-specific special conditions and grant requirements. Research grantees should work with their assigned OJJDP program manager on specific questions, but this site outlines the general policies and procedures of those conditions.
Review slides from a previous New Grantee Orientation. Access the PowerPoint presentation slides.
Quick Links
- Privacy and Human Subjects Protections
- Full and Complete Documentation for Privacy Certificate Compliance
- Full and Complete Documentation for Human Subjects Compliance
- Research and Evaluation Independence and Integrity
- Press Releases and Publications
- Final Reports and Documentation for Research Grantees
- Copyright, Data Rights, and Data Archiving
- Key Personnel
- Progress Reporting
- Introduction to Reports
- Goals and Accomplishments
- Products and Deliverables
- Participants and Other Collaborating Organization
- Impacts or Contributions to the Field
- Changes, Problems and Solutions
- Next Steps
- Budget and Other General Grant Requirements and Resources
Privacy and Human Subjects Protections
Department of Justice (DOJ) regulations (28 C.F.R. Part 22) require recipients of OJP funding to submit a Privacy Certificate as a condition of approval of any grant application or contract proposal that contains a research or statistical component under which "information identifiable to a private person" will be collected, analyzed, used, or disclosed.
DOJ regulations (28 C.F.R. Part 46) protect the human subjects of federally funded research. Part 46 requires that an Institutional Review Board (IRB), in accordance with the regulations, review and approve most research involving human subjects that any federal department or agency conducts or supports before an award recipient may expend federal funds for that research.
OJJDP applies these regulations to all grant awards involving research through the following withholding special condition:
The recipient will be permitted to obligate, draw down, or expend up to [$XX,XXX] in funds upon award to engage in activities that do not constitute research involving human subjects or the collection of information identifiable to a private person. The recipient will not be permitted to obligate, expend or draw down any part of the remainder of awarded funds for any research or statistical activity or project involving the collection, use, analysis, transfer, or disclosure of information identifiable to a private person until:
- the recipient has submitted full and complete documentation to demonstrate that it will conduct or perform research involving human subjects in accordance with an approved Federal-wide assurance issued by HHS;
- the research has been determined, by an appropriate IRB to be an exempt research activity, or has been reviewed and approved by an appropriate IRB in accordance with the requirements of 28 CFR Part 46;
- the recipient has submitted a properly executed Privacy Certificate in accordance with the requirements of 28 CFR Part 22;
- an OJP Human Subjects Protection Officer has reviewed the documentation submitted and has authorized, in writing, removal of this special condition; and
- a Grant Adjustment Notice has been issued removing this special condition.
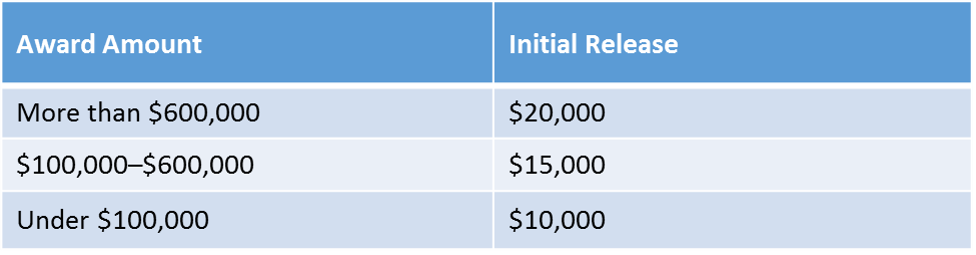
The amount of the withholding special condition depends on the amount of the award as illustrated in the following chart:
In order to remove the withholding special condition, grantees need to submit full and complete documentation for Privacy and Human Subjects Compliance outlined below. These documents should be submitted as "special reports" for OJJDP review. The documentation is not considered approved until a separate grant adjustment notice has been fully approved to lift this special condition.
Full and Complete Documentation for Privacy Certificate Compliance
- A completed privacy certificate must include a description of the grantee's policies and procedures to protect the confidentiality of identifiable data (demonstrating compliance with 28 C.F.R. Part 22).
- The privacy certificate must include the signature and names of the (1) principal investigator/co-investigators (if applicable), and (2) authorized institutional representative, who is authorized to sign on behalf of the granted agency. There should be at least two signatures on the privacy certificate.
- The privacy certificate should indicate that de-identified data will be archived at the National Archive of Criminal Justice Data at the conclusion of the project.
- If the study is not collecting or using personally identifiable information, grantees should state this explicitly using the following statement: "No data identifiable to a private person will be collected," and complete the form using the terms N/A or Not Applicable including a brief description of why the particular item is not applicable.
- Upload modifications to privacy certificates throughout the award as special reports in the Grants Management System as soon as possible after the modification is in place, but, at minimum, at the same time as the progress report.
Full and Complete Documentation for Human Subjects Compliance
- Completed Human Subjects Form with the IRB review determination, reference to the review being conducted according to 28 C.F.R. Part 46, and is signed by an IRB Official;
Guidance for OJJDP Research Grantees
OR
- A letter, on institutional letterhead, signed by the IRB chairman that:
- Includes the Federal Wide Assurance (FWA) number.
- Includes the IRB review determination.
- References 28 C.F.R. Part 46 (in addition to or instead of 45 C.F.R. Part 46).
- Renewal or expiration date of the IRB determination.
Guidance for OJJDP Research Grantees
AND
The IRB-reviewed and approved or exempted protocol. The protocol must include:
- Includes as an attachment the IRB-reviewed and approved or exempted protocol, including all consent forms.
- Any application materials or research methods description submitted to the IRB;
- All informed consent and assent forms for research involving human subjects; and
- Explicit description of any planned research incentives (if proposed).
- A Research Subject Incentive and Support Cost Approval Request (if proposed).
The grantee should upload renewals and amendments as special reports in the Grants Management System as soon as possible after the IRB approves the modification or renewal, but, at minimum, at the same time as the progress report.
Confidentiality of Program Records: For projects that do not involve a research component, see the following section of the Juvenile Justice and Delinquency Prevention Act pertaining to the confidentiality of program records:
Except as authorized by law, program records containing the identity of individual juveniles gathered for purposes pursuant to this title may not be disclosed without the consent of the service recipient or legally authorized representative, or as may be necessary to carry out this title. Under no circumstances may program reports or findings available for public dissemination contain the actual names of individual service recipients.
Research Participant Support Costs and Incentives
Research participant support costs, including stipends, for participation in research activities and incentives for human subjects in research studies that are reasonable, allocable, and necessary for the project and are consistent with the DOJ Financial Guide are allowable for some OJJDP-funded social science research. The following outlines the protocols for OJJDP review and approval of two types of these costs: (1) research support costs for participants who are not human subjects and (2) research incentives and support costs for human subjects.
1. Research support costs for participants who are not human subjects. These generally cover participants' direct costs in a research study, including subsistence allowances, travel allowances, and registration fees paid to or on behalf of participants or trainees the study who are not human subjects (or employees). Participant stipends, a type of participant support cost, are also outlined below.
- Along with the other costs in the proposed budget, OJJDP and the Office of Justice Programs' Office of the Chief Financial Officer staff review any participant support costs in the budget to assess whether they are reasonable, allocable, and necessary for project activities.
- Applicants should label these types of costs as "research participant support costs" in the budget and, as with other cost items, the budget should contain all calculations and appropriate narrative explaining and justifying the costs. The budget narrative should explain:
- The total amount of the research participant support costs.
- The per capita calculation (i.e. cost per staff person, site, etc.), including a breakdown by tasks if multiple payments are involved (e.g., several interviews/surveys over time).
- The specific form the support costs will take, such as a stipend or other costs (food, beverages, raffles, entertainment, and lotteries are not allowed in any form).
- Justification that the amount of compensation is reasonable/nominal. Compensation must be modest so as not to be out of proportion to time and effort involved in the study.
- How the grantee will distribute and track support costs.
- After reviewing this information, OJJDP and the Office of the Chief Financial Officer typically approve research participant support costs as part of the review and approval process for the full grant award budget.
- Grantees must have a disbursement policy and a tracking system in place before they disburse participant support costs.
- Research participant stipends are a type of support cost defined as payments (including cash) to study participants who are acting in their professional role (e.g. teachers filling out student assessments, data analysts retrieving administrative records, etc.). They can also be applied to institutions, such as schools, to reimburse them for costs associated with participation in studies, such as space, staff time costs, supplies, etc. Stipends can include reimbursement to study participants for expenses incurred related to travel, lost work time, etc. (However, if the participants are human subjects, then grantees must follow the procedure for human subjects below.)
- If proposed, stipends must reflect the time and effort of participation and be directly related to the costs of participation.
- Research stipends must also be limited to time spent that is outside of the normal working hours of the professional contributing to the study. Individuals cannot be paid double for working within their normal paid working hours.
2. Research incentives and support costs for human subjects. An incentive is defined as a positive motivational influence, something that induces action or motivates effort. Researchers sometimes use incentives to encourage participation beyond just the intrinsic value of being part of the research study. Incentives may be monetary or nonmonetary, such as books, calculators, etc.
Incentives are most appropriately used with hard-to-find populations or respondents whose failure to participate would jeopardize the quality of the survey data (e.g., in surveys experiencing high attrition) or in studies that impose exceptional burdens on respondents, such as those asking highly sensitive questions.
Research support costs for human subjects are generally the same types of costs outlined in the section above (Research support costs for participants who are not human subjects); but, are provided to human subjects in a study.
Under certain circumstances related to participation in research studies, OJJDP may approve the use of incentives and other research support costs for human subjects, provided they are reasonable and in proportion to the time and effort involved in study participation.
Research grantees may offer incentives or support costs to human subjects only after OJJDP's human subjects protection officer and Deputy Associate Administrator of the Innovation and Research Division fully approve a "Research Incentive and Support Cost Approval Request" and the Full and Complete Documentation for Human Subjects Compliance and the grant adjustment notice to lift the associated withholding condition has been fully approved. The human subject participant costs and incentives must also be consistent with the DOJ Financial Guide and be approved as part of the grant award budget in the "other" category.
Grantees who feel human subject incentives and support costs are critical to the integrity of the study must submit a "Research Subject Incentive and Support Cost Approval Request" to OJJDP as part of Full and Complete Documentation for Human Subjects Compliance. This request should include:
- A description of the costs that includes:
- The total amount of the incentive or support cost, including the amount to be provided per respondent and a breakdown by tasks if multiple payments are involved (e.g., several interviews/surveys over time).
- The specific form the incentives or support costs will take (food, beverages, raffles, entertainment, and lotteries are not allowed in any form).
- Justification that the amount of compensation is reasonable/nominal. Compensation must be modest so as not to be out of proportion to time and effort involved in the study.
- How the grantee will distribute and track the incentives or costs.
- A justification of the need for the incentives or support costs that includes:
- Evidence that the use of incentives is justified to recruit subjects to the study or to keep subjects involved in a study across multiple points of data collection. This evidence should be study-specific.
- The estimated impact if incentives were not used.
- An explanation of the potential human subjects considerations that includes:
- The date the Institutional Review Board approved the support cost or incentive amounts. OJJDP will not approve incentives without documentation of the prior approval of the Institutional Review Board, which the grantee must also upload into the Grants Management System.
- A description of the age range and financial status of participants receiving incentives or costs (in particular, if students/youth and low-income individuals are involved).
- How the grantee will eliminate or minimize the possibility of undue influence or coercion rather than voluntary participation for study subjects.
Research and Evaluation Independence and Integrity
OJJDP requires grantees conducting research to comply with the Office of Justice Programs' (OJP) Research and Evaluation Independence and Integrity (REII) Policy. Recipients of OJP funding for research activities must demonstrate (1) personal, (2) financial, and (3) organizational independence and integrity regarding both this proposed research and/or evaluation and any current or prior related projects. OJP will assess research and evaluation independence and integrity based on considerations, such as the adequacy of the applicant's efforts to identify factors that could affect the objectivity or integrity of the proposed staff and/or the applicant entity (and any subrecipients) in carrying out the research, development, or evaluation activity; and the adequacy of the applicant's existing or proposed remedies to control any such factors.
If a grantee's REII documentation is insufficient, OJJDP will place a withholding special condition on the research project's grant award. Once a grantee's REII documentation is addressed and found to be sufficient, a grant adjustment notice will be approved by OJJDP to lift the RII special condition.
Required Documentation:
Each research grant award recipient must submit to OJJDP one of the following two items that document research and evaluation independence and integrity:
- A specific assurance that the applicant has reviewed its application to identify any actual or potential apparent conflicts of interest (including through review of pertinent information on the principal investigator, any coprincipal investigators, and any subrecipients), and that the applicant has identified no such conflicts of interest-whether personal or financial or organizational (including on the part of the applicant entity or on the part of staff, investigators, or subrecipients)-that could affect the independence or integrity of the research, including the design, conduct, and reporting of the research.
If an applicant reasonably believes that no actual or potential apparent conflicts of interest (personal, financial, or organizational) exist, then the applicant should provide a brief narrative explanation of how and why it reached that conclusion. The applicant must also include an explanation of the specific processes and procedures that the applicant has in place, or will put in place, to identify and prevent (or, at the very least, mitigate) any such conflicts of interest pertinent to the funded project during the period of performance. Documentation that may be helpful in this regard may include organizational codes of ethics/conduct and policies regarding organizational, personal, and financial conflicts of interest. There is no guarantee that the plan, if any, will be accepted as proposed.
- A specific description of actual or potential apparent conflicts of interest that the applicant has identified-including through review of pertinent information on the principal investigator, any coprincipal investigators, and any subrecipients-that could affect the independence or integrity of the research, including the design, conduct, or reporting of the research. These conflicts may be (1) personal (e.g., on the part of investigators or other staff), (2) financial, or (3) organizational (related to the applicant or any subrecipient entity). Some examples of potential investigator (or other personal) conflict situations are those in which an investigator would be in a position to evaluate a spouse's work product (actual conflict), or an investigator would be in a position to evaluate the work of a former or current colleague (potential apparent conflict). With regard to potential organizational conflicts of interest, as one example, generally an organization would not be given an award to evaluate a project if that organization had itself provided substantial prior technical assistance to that specific project or a location implementing the project (whether funded by OJP or other sources), because the organization in such an instance might appear to be evaluating the effectiveness of its own prior work. The key is whether a reasonable person understanding all of the facts would be able to have confidence that the results of any research or evaluation project are objective and reliable. Any outside personal or financial interest that casts doubt on that objectivity and reliability of an evaluation or research product is a problem and must be disclosed.
If the applicant has identified actual or potential apparent conflicts of interest (personal, financial, or organizational) that could affect the independence and integrity of the research, including the design, conduct, or reporting of the research, the applicant must provide a specific and robust mitigation plan to address each of those conflicts. At a minimum, the applicant is expected to explain the specific processes and procedures that the applicant has in place, or will put in place, to identify and eliminate (or, at the very least, mitigate) any such conflicts of interest pertinent to the funded project during the period of performance. Documentation that may be helpful in this regard may include organizational codes of ethics/conduct and policies regarding organizational, personal, and financial conflicts of interest. There is no guarantee that the plan, if any, will be accepted as proposed.
Press Releases and Publications
Press Releases: OJJDP research grantees shall transmit to OJJDP copies of all official award-related press releases at least 5 working days prior to public release. Advance notification allows OJJDP time to coordinate the release of information, where appropriate, and to respond to press or public inquiries.
Publications: OJJDP generally expects the award recipient to prepare publications, which include any planned written, visual, or sound materials substantively based on the research project, for dissemination to the public. This expectation also generally includes scholarly publications, such as peer-reviewed, scientific journal articles and/or book chapter(s) or book(s) in the academic press. When publishing products developed with and/or about studies funded with OJJDP grant funds, OJJDP expects grantees to:
- Notify OJJDP if a scholarly product has been accepted at least 3 weeks in advance of publication. This notification should include: (1) the anticipated date of publication; (2) the title of the publication; (3) the publisher and location (online or city); (4) the authors; and (5) a brief abstract.
OJJDP encourages grantees to submit independent publications to OJJDP in advance of finalization for review and comments.
- Include a disclaimer on publications that indicates: "This project was supported by Grant #___ awarded by the Office of Juvenile Justice and Delinquency Prevention, Office of Justice Programs, U.S. Department of Justice. The opinions, findings, and conclusions or recommendations expressed in this publication/program/exhibition are those of the author(s) and do not necessarily reflect those of the Department of Justice." Include this disclaimer on all final technical reports.
- Submit publications developed with grant funds that OJJDP publishes (such as those published on OJJDP.gov and NCJRS.gov or through the OJP or OJJDP publications process) or those that include an OJJDP seal or logo to OJJDP in advance for research and communications staff review and approval.
- Submit a final technical report for all research grant awards to OJJDP.
Final Reports and Documentation for Research Grantees
At the conclusion of a grant award, OJJDP expects final reports to be submitted through JustGrants. For OJJDP research grantees, this includes four types of reports or documentation that should be uploaded:
- The final IRB review documentation and privacy certificates, which include final approvals, modifications, and packages submitted for review, if not submitted to OJJDP previously. These should be uploaded and submitted in a separate special report.
- A final progress report that summarizes the extent to which the goals and objectives were met throughout the award period. See Progress Reporting Template for more information. This should be uploaded as one of at least three attachments in the final report.
- A final technical report that summarizes the research questions or objectives, methods, and analytical techniques of the study and the findings and conclusions, similar to the type of information reported in a refereed journal. This should be uploaded as the second of at least three attachments in the final report. The final technical report should address each of the items in the Final Technical Report Outline.
OJJDP social science analysts review all final technical reports for completeness and clarity and offer recommendations regarding technical edits or additional content. Once the final report is approved, OJJDP provides a copy for public access on the OJJDP and Office of Justice Programs websites.
In addition, OJJDP notifies the Attorney General of the pending publication through an entry in a weekly memo. Publications generally need to appear in this weekly notification at least twice prior to the publication being available to the public.
- A plain language summary of the final technical report that will summarize the project research findings in plain, practice-oriented language that will facilitate understanding and application. These summaries should follow OJJDP's template for creating the summaries. This should be uploaded as the third of at least three attachments in the final report. These will be converted into a standard template to be included with the final technical report on NCJRS.
Because the social science analyst review of final technical reports and summaries often result in requested or suggested edits, grantees should submit a complete draft of both documents to the OJJDP social science analyst 60 days before the end of the project period. The final versions, submitted within 30 days of the conclusion of the project period, should address all the edits, as necessary.
At the conclusion of the project, grantees should archive all data at the National Archive of Criminal Justice Data.
Copyright, Data Rights, and Data Archiving
Copyright: OJP reserves a royalty-free, nonexclusive, and irrevocable license to reproduce, publish, or otherwise use for federal purposes: (1) any work subject to copyright developed under an award or subaward and (2) any rights of copyright to which a recipient or subrecipient purchases ownership with federal support.
Data Rights: OJP has the right to (1) obtain, reproduce, publish, or otherwise use the data first produced under an award or subaward and (2) authorize others to receive, reproduce, publish, or otherwise use such data for federal purposes.
Data Archiving
As of FY 2016 (and for select grantees in prior years), all research grantees are required to deliver a public-use dataset containing all data collected under this project 30 days before the end of the project period to the National Archive of Criminal Justice Data unless OJJDP notifies the grantee in writing that they are exempt from this requirement. Archiving includes the submission of the following files and materials:
- The data files of the de-identified data collected under the study
- Technical documentation, which includes:
- Codebooks
- Syntax or special coding
- User guide
- Study documentation, which includes:
- IRB approval
- Informed consent
- OJJDP approved privacy certificate
- OJJDP approved data archiving plan
- Final report
- Bibliographies.
To ensure that the data will be archived at the conclusion of the projects, OJJDP expects grantees to:
- Submit to OJJDP a data archiving plan, with specific schedule, tasks, and milestones, within 6 months of the grant period start date. This should be uploaded as a special report.
- Throughout the project, the recipient will make no guarantee, without prior OJJDP approval, that the data collected, acquired, or produced as part of this project will not be transferred or released. For example, such a guarantee in an informed consent form would preclude OJJDP from archiving and making available OJJDP-funded data.
- The privacy certificate should also include reference to archiving the data at the National Archive of Criminal Justice Data.
Also, review the OJJDP research grantees presentation on the Archiving Orientation.
Key Personnel
The grantee may replace the project director (or project managers) and key program personnel designated in the application (generally principal investigators) only under compelling reasons. OJJDP must approve successors to key personnel, and such approval is contingent upon the grantee submitting appropriate information, including, but not limited to, a resume. OJP will not withhold approval unreasonably. Changes in other program personnel require only notification to OJP and submission of resumes, unless otherwise designated in the award document.
Progress Reporting
OJJDP requires that grantees submit to OJJDP every 6 months reports that detail the progress they have made during the reporting period. A template of the elements that research project progress reports should address is available:
Reporting periods:
- January 1 - June 30: Due July 30
- July 1 - December 31: Due January 30
Research Award Closeout
The project period end date is the final date that the grantee can incur costs. (Grantees generally may request one no cost extension for as long as 12 months. See the DOJ Financial Guide.)
OJJDP research grantees should start to prepare for closing out the grant award at least 3 months prior to the end date of their award. Grantees should start the formal closeout process as soon as the program is completed and/or they have spent all federal and matching funds.
Key closeout steps include:
- 60 days prior to the project period end date: Submit a draft final technical report and plain language summary to OJJDP for social science analyst review and comments.
- 30 days prior to the project period end date: Submit all data and associated documentation to the National Archive of Criminal Justice Data.
- At the project period end date:
- The formal closeout period begins.
- The grant can no longer incur or obligate expenses.
- 30 days after the project period end date:
- Upload the final progress report, final technical report, and plain language summary and the final report. These should address the social science analyst comments.
- Upload the final IRB approvals and associated documentation and the final privacy certificate as special reports.
- No later than 90 days after the project period end date:
- Submit the final financial status report.
- Complete the programmatic requirements checklist.
- Submit the closeout package.
- After the project is closed:
- Continue to protect the confidentiality and privacy of human subjects.
- Continue to notify OJJDP of publications about the research.
Budget and Other General Grant Requirements and Resources
OJJDP research grantees also must follow the standard OJP grant policies and procedures. Additional resources include:
- Budget Detail Worksheet Template
- DOJ Grants Financial Guide
- All points of contact and financial points of contact are required to complete the Financial Management Training.
- OJP Grantee Resources
- For financial questions, please contact the Office of Chief Financial Officer Helpdesk: [email protected]; 800–458–0786
Introduction to Reports
- Federal Agency and Organization Element to Which Report is Submitted
- Federal Grant or Other Identifying Number Assigned by Agency
- Project Title
- PD/PI Name, Title and Contact Information (e-mail address and phone number)
- Submission Date
- Project/Grant Period (Start Date, End Date)
- Reporting Period End Date
- Report Term or Frequency (annual, semi-annual, quarterly, special, or other)
- Current IRB Approval/Renewal/Modification and Expiration Date (Please then attach the associated approval letters, submission materials, and description of the changes if renewed or modified during the reporting period).
Goals and Accomplishments
- What are the major goals and objectives of this project?
- What was accomplished under these goals?
- What opportunities for training and professional development has the project provided?
- How have the results been disseminated to communities of interest?
Products and Deliverables
- What has the project produced? (Please attach copies, as applicable)
Participants and Other Collaborating Organization
- What individuals have worked on the project?
- What other organizations have been involved as partners?
- Organization Name
- Location of Organization
- Partner's Contribution
- Have other collaborators or contacts been involved?
Impacts or Contributions to the field
- Describe distinctive contributions, major accomplishments, innovations, successes, or any change in practice or behavior that has come about as a result of the project.
Changes, Problems and Solutions
- Changes in approach and reasons for change.
- Actual or anticipated problems or delays and actions or plans to resolve them and a modified timeline, when appropriate.
- Changes that have a significant impact on expenditures.
- Changes to Key Staff.
Next Steps
- What do you plan to do during the next reporting period?